Five-year project and strategy
Syensqo has recently announced its new GROW strategy and reorganized its business into three segments: Materials, Chemicals, and Solutions with different priorities and needs in terms of innovation. With increasing concerns about climate change and the societal impact of chemical companies, sustainable solutions and the use of biomass-derived raw materials have become key business drivers. In parallel, digital technologies, in particular, Artificial Intelligence (AI) and data analytics, are redefining the way of doing research, and have been identified by Syensqo and the CNRS as key priorities to remain at the cutting edge of innovation.
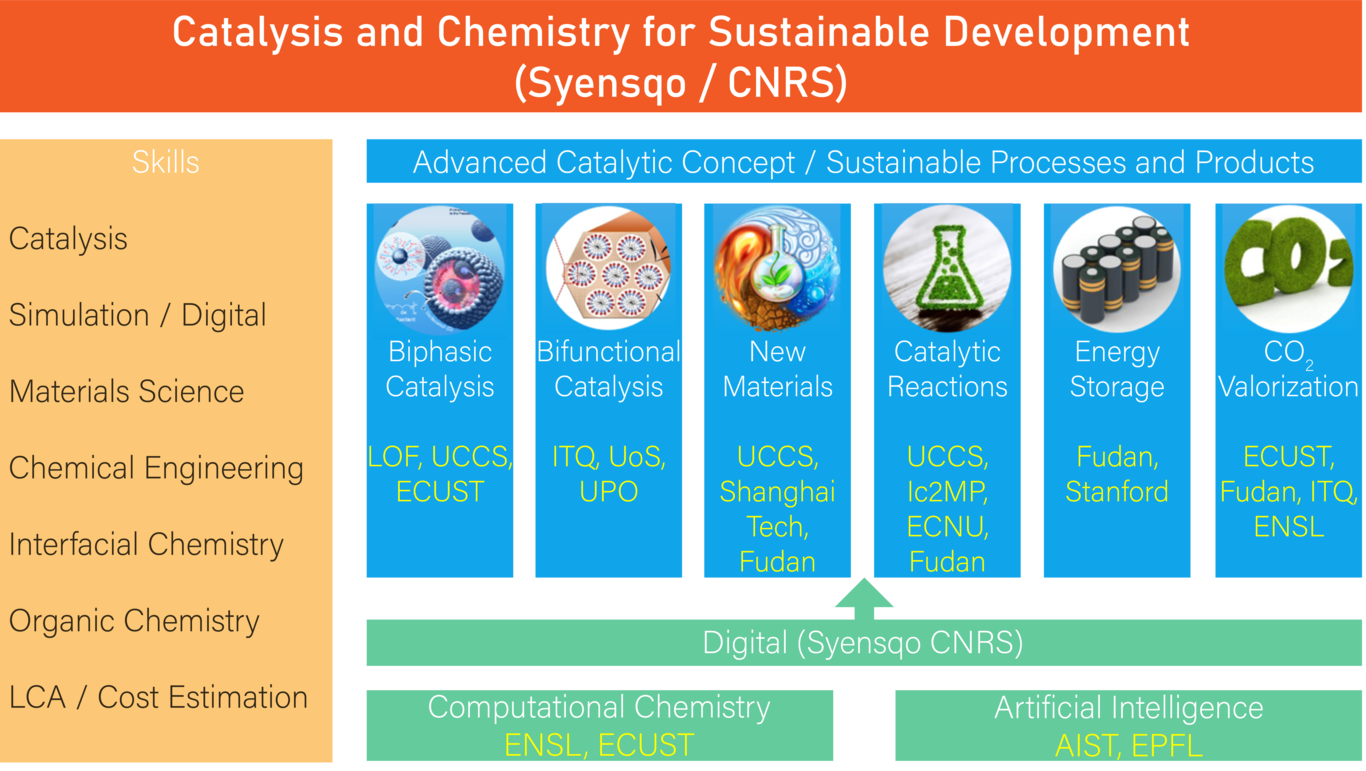
Within this context, and building on experience collected over the last years in working with GBUs, we have decided to kick off an ambitious “digital catalysis roadmap” and to refocus our projects into two main research lines to deliver more value to Solvay, while ensuring a high scientific level.
The main objective of the platform will be the development of sustainable catalytic solutions (amination-, oxidation-, hydrogenation-, electro-catalysis) to replace or improve existing processes of Syensqo's GBUs in order to increase the sustainability. In addition, we will design new routes to existing products, starting from biomass or CO2. Its second objective will be the development of new, sustainable products enabling, for instance, the storage of renewable energy. All projects will be carried out in collaboration with the strong network of E2P2L and dedicated collaborations, as necessary.